Helicobacter pylori infection is a bacterial disease of the stomach.
Helicobacter pylori infection can be symptomatic and asymptomatic. Marshall and Warren were awarded the Nobel Prize in 2005 for the discovery of the bacterium of the same name in 1983.
Definition
Helicobacter pylori is a gram-negative rod-shaped bacterium with lophotrichous flagella that can colonize the human stomach and cause gastritis. In this case, it is referred to as an infection and a disease, regardless of whether symptoms occur or not. Helicobacter pylori infection is associated with other secondary diseases such as peptic ulcer disease, adenocarcinoma of the stomach or marginal zone B-cell lymphoma of the MALT (mucose-associated lymphoid tissue).
Epidemiology
The global prevalence of Helicobacter pylori infection (H. pylori) varies greatly between different regions. It is highest in Africa at 70.1%, with a minimum prevalence of 24.4% in Oceania. Within Europe, the prevalence is variable, with a maximum of 86.4% in Portugal and a minimum of 18.9% in Switzerland. H. pylori is considered a major risk factor for the development of peptic ulcer disease and gastric carcinoma. Depending on the area, around 60% of ulcers are etiologically attributable to H. pylori infection, while in around 90% of cases H. pylori infection is considered to be the cause of gastric carcinoma.
Causes
The exact mode of transmission of H. pylori infection is still unclear. Infection within the family is considered to be a significant factor in the transmission of infection. Possible transmission routes are oral-oral, gastral-oral and fecal-oral spread or a combination of these.
Pathogenesis
H. pylori colonizes the mucosa of the stomach and has the ability to produce urease. This is an enzyme that breaks down urea into carbon dioxide and ammonia. Ammonia increases the pH value in the immediate vicinity of the bacterium, protecting it from hydrochloric acid. Other enzymes secreted by H. pylori, such as protease, lipase and phospholipase A2, inhibit the production of mucus, which protects the epithelium against the acidic gastric environment. The resulting disintegration of the mucosal layer allows the bacterium to bind specifically to gastric epithelial cells.
This binding provides H. pylori with mechanical protection against gastric motor activity and also triggers an immune response from the host cells. This results in further damage to the gastric mucosa, as cytotoxic T cells can induce apoptosis by recognizing antigen-active peptides on the surface of epithelial cells.
The permanent disruption and inflammation of the gastric mucosa caused by these pathways is known as chronic gastritis, which is asymptomatic in most people. In some cases, the altered gastric secretion in combination with tissue damage can lead to the development of peptic ulcer disease. In addition, cell atrophy, interstitial metaplasia and finally gastric carcinoma can develop.
The CagA gene (cytotoxin-associated gene A), which codes for a protein of the same name, can be detected in almost all H. pylori strains isolated in East Asia, for example, while it is absent in around 30-40% of strains isolated in Western Europe. The protein causes improved permeability of the epithelium for urea, so that more substrate is available to the bacterial urease and the infectivity of the pathogen is increased. CagA-positive strains are therefore considered to have an increased risk of developing peptic ulcers. Furthermore, CagA-positive strains have been associated with an increased risk of precancerous lesions and gastric carcinomas for many years. Recent studies have shown that the CagA protein inhibits tumor suppressor genes.
Symptoms
The clinical manifestation of an H. pylori infection is very variable. Most of those affected are asymptomatic, while symptomatic cases may present with non-specific mild gastrointestinal complaints and even acute bleeding and perforation as ulcer complications. As the H. pylori infection leads to inflammation of the gastric mucosa, the most common symptoms manifest themselves in the context of acute gastritis. There is no typical symptom pattern for this. Possible symptoms are
- A feeling of fullness
- Heartburn
- Pain in the upper stomach
- nausea
- Vomiting
In chronic gastritis, these symptoms may occur in a minor form or be absent. However, it is not possible to differentiate between H. pylori-positive and H. pylori-negative gastritis solely on the basis of the clinical symptoms.
Diagnostics
It is only recommended to test patients for the presence of H. pylori if a positive test would influence treatment. Indications for testing are
- History of known or newly detected ventricular or duodenal ulcer
- Gastric MALT lymphoma
- Dyspepsia
- ITP (idiopathic thrombocytopenic purpura)
- IgA vasculitis
- Unexplained or refractory iron deficiency anemia
- Increased risk of gastric carcinoma
- Planned long-term therapy with low-dose acetylsalicylic acid (ASA), non-steroidal anti-inflammatory drugs (NSAIDs) and/or anticoagulation
- in patients with an increased risk of ulcer disease
- Development of gastroduodenal bleeding while taking NSAIDs, ASA, anticoagulants and/or P2Y12 inhibitors
In general, the diagnosis of an H. pylori infection is considered reliable if there are two positive results with different test methods, although this rarely occurs in everyday practice. In the following constellations, a second test procedure is not necessary:
- Histological evidence of H. pylori is present in combination with chronic active gastritis
- There is a duodenal ulcer and a positive test result for H. pylori with a high H. pylori prevalence
Under these circumstances, an H. pylori infection is considered confirmed. It should be noted that a negative culture does not rule out H. pylori infection and requires further diagnostics if clinical suspicion persists.
Urease breath test: Urease breaks down urea into carbon dioxide and ammonia. Bacterial overgrowth in the stomach can lead to false-positive results, as there are other bacteria besides H. pylori that break down urea. Pre-treatments with proton pump inhibitors (PPI), recent antibiotic therapies and bleeding of the upper gastrointestinal tract can simulate false negative results.
Rapid antigen tests: Other non-invasive test methods include rapid stool antigen tests based on monoclonal antibodies and IgG antibody tests to detect antibodies in serum.
Invasive procedures: The invasive tests are based on an endoscopic procedure with biopsy removal. The tissue samples are then examined histologically or subjected to a rapid urease test. Histology is the most informative method, as the histological inflammation pattern of chronic gastritis in combination with the bacterial morphology can reliably detect an H. pylori infection and also provide information on the relevance of treatment, prognosis and the risk of developing a carcinoma. If symptoms are present that indicate damage to the upper gastrointestinal tract, endoscopic testing with biopsy is justified. Histological diagnosis is also recommended if there is a history of partial gastric resection. Under these circumstances, the surface area of the gastric mucosa is reduced, so that the sensitivity of non-invasive tests is impaired. Histology is also preferred in the case of PPI therapy, as the bacterial density is reduced in this case and the sensitivity of non-invasive tests is impaired. In this context, PPI therapy should be paused for two weeks prior to diagnosis, if clinically justifiable, and no antibiotic therapy should be given in the preceding four weeks.
Differential diagnosis
In addition to H. pylori gastritis, there are many other types of gastritis with a different etiology, such as chemically-induced/reactive gastritis or autoimmune gastritis. As the symptoms of (H. pylori) gastritis are non-specific, it is important to consider other diseases of the gastrointestinal tract in the differential diagnosis:
- Crohn's disease
- GERD (gastroesophageal reflux disease)
- Gastroduodenal ulcer: In addition to an H. pylori infection, other factors can trigger the development of an ulcer, such as alcohol and nicotine consumption, NSAID use, stress, genetic predisposition, Zollinger-Ellison syndrome, gastric adenocarcinoma
What treatment helps with Helicobacter pylori infection?
The aim of treating an H. pylori infection is to eradicate the pathogen. This is achieved with medication.
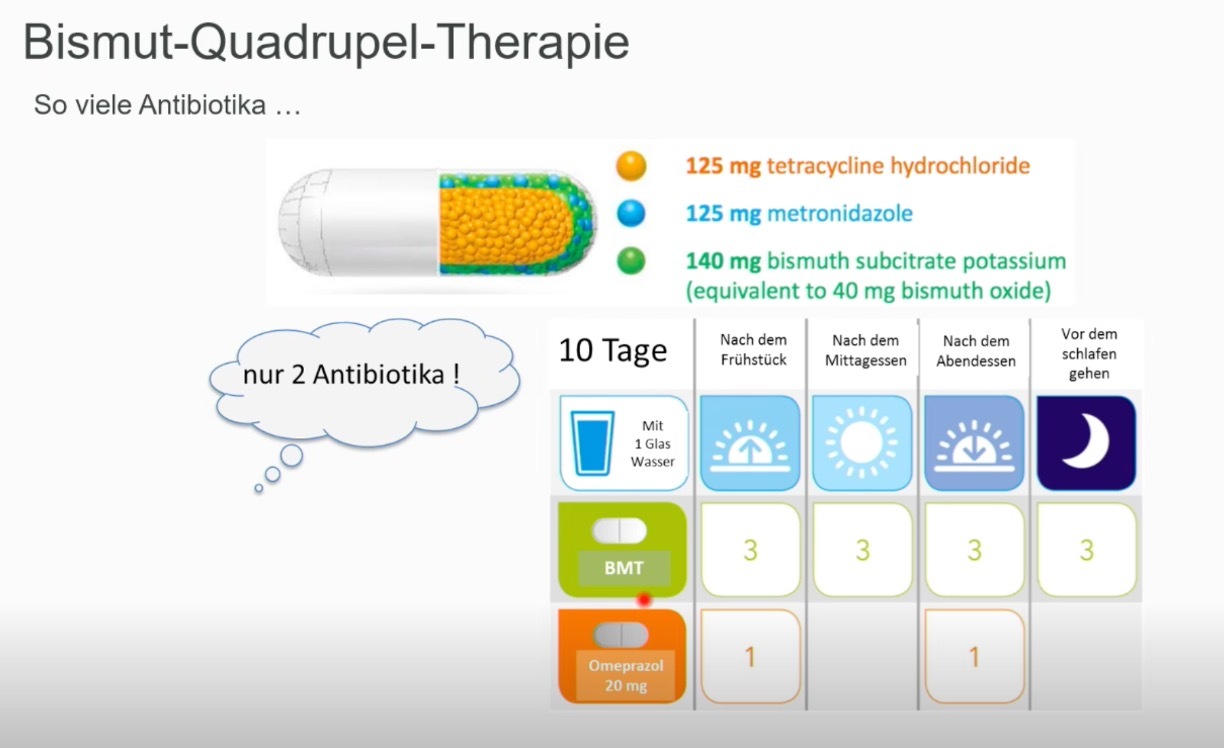
Bismuth-containing quadruple therapy for ten days is recommended as first-line therapy, with the aim of achieving an eradication rate of >90%. This quadruple therapy consists of proton pump inhibitors, bismuth, tetracycline and metronidazole (Pylera as a combination preparation).
If first-line therapy is unsuccessful, resistance testing is recommended. In particular, possible resistance to clarithromycin should be investigated, as this is a key component of the standard triple therapy, which is recommended as a second-line therapy for 14 days. It consists of proton pump inhibitors, clarithromycin and amoxicillin.
Pre-therapeutic resistance to amoxicillin is very rare. If there is a penicillin allergy, amoxicillin can be replaced by metronidazole. Triple therapy containing fluoroquinolones can also be used. If the second-line therapy also fails, further therapy attempts are made exclusively by specialists according to reserve regimens.
Stopping smoking helps to improve the effectiveness of the therapy. The success of the therapy is checked at least four weeks after the end of antibiotics. Routine follow-up checks to rule out H. pylori reinfection are not recommended.
Helicobacter pylori gastritis: eradication with bismuth (Pylera).
Eradication scheme Helicobacter
First-line treatment:
- Clarithromycin 2x500 mg/day, amoxicillin 2x1000 mg/day, PPI for 14 days
- Pylera 140 mg/125 mg/125 mg hard capsules 4x3 capsules/day (postprandial), PPI 2x40mg pantoprazole equivalents for 10 days: Please keep a time interval of at least 3 hours between the consumption of calcium-containing foods (e.g. milk, yoghurt, quark, cheese, calcium-containing drinking water) and the intake of the drug.
First-line treatment for penicillin allergy:
- Clarithromycin 2x500 mg/d, metronidazole 2x500 mg/d, PPI for 14 days
- Pylera 140 mg/125 mg/125 mg hard capsules 4x3 capsules/day (postprandial), PPI 2x40mg pantoprazole equivalents for 10 days: Please keep a time interval of at least 3 hours between the consumption of calcium-containing foods (for example milk, yoghurt, quark, cheese, calcium-containing drinking water) and the intake of the drug.
Second-line treatment:
- Pylera 140 mg/125 mg/125 mg hard capsules 4x3 capsules/day (postprandial), PPI 2x40mg pantoprazole equivalents for 10 days: please maintain a time interval of at least 3 hours between consuming foods containing calcium (for example milk, yogurt, curd, cheese, drinking water containing calcium) and taking the medicine.
- Levofloxacin 2x500 mg/day, amoxicillin 2x1000 mg/day for 14 days
Third-line treatment:
Third-line treatment must be planned individually on the basis of a prior antibiogram.
How is treatment monitoring (eradication success documentation) carried out after HP infection?
Eradication success monitoring after Helicobacter pylori treatment refers to checking whether the treatment carried out was successful and whether the Helicobacter pylori (H. pylori) bacterial strain has been completely eliminated from the patient's gastrointestinal tract. This is done by a special examination about 4 weeks after the end of the antibiotic treatment. As a rule, a stool sample (Hp stool test) is used to search for remnants of H. pylori. The eradication success control serves several purposes:
Ensuring the effectiveness of the treatment: the check ensures that the therapy used has achieved the desired results and that the bacteria have been effectively eliminated.
Prevention of relapses: A successful eradication outcome minimizes the risk of relapse and re-infection with H. pylori, which is important to prevent long-term health problems.
Prevention of antibiotic resistance: Monitoring helps to ensure that the antibiotics used are still effective. Incomplete treatment could lead to antibiotic resistance, which could make future treatment more difficult.
It is important to follow the doctor's instructions exactly and to carry out the prescribed eradication success check to ensure that the H. pylori infection has been successfully treated and to prevent any complications.
What are complications of Helicobacter pylori infection?
Possible complications and secondary diseases of an H. pylori infection are
- Gastroduodenal ulcer diseaseAdenocarcinoma of the stomachMALT lymphoma
What is the prognosis of a Helicobacter pylori infection?
H. pylori infection always leads to chronic gastritis. About 10% of infected persons develop ulcers, while in about 1% the disease progresses to gastric carcinoma if left untreated. Reinfection after successful eradication is rare and occurs in 2% of cases per year in industrialized countries, compared to 6-12% in developing countries.
What can be done to prevent Helicobacter pylori infection?
For all people with an increased risk of gastric cancer, it is recommended that endoscopic biopsy testing for H. pylori is carried out from the age of 40 and that eradication therapy is started if the result is positive. In addition to preventing gastric cancer, this contributes to the prevention of other gastroduodenal diseases.