Ulcerative colitis, a chronic inflammatory bowel disease (IBD).
Ulcerative colitis is a form of chronic inflammatory bowel disease that mainly affects the mucous membrane in the large intestine.
Ulcerative colitis is often accompanied by bloody diarrhea caused by inflammation and ulcers in the intestinal mucosa. Typical symptoms are
- persistent diarrhea,
- Abdominal pain,
- Blood in the stool
- Weight loss.
- frequent bowel movements
- Feeling of urgency (urgency)
The disease progresses in waves: phases with symptoms (flare-ups) and periods without symptoms (remission).
In addition to bowel complaints, other symptoms may also occur, such as joint inflammation. People with this disease have an increased risk of developing bowel cancer. Diagnosis is usually made by colonoscopy and treatment includes various medications such as 5-aminosalicylic acid, corticosteroids, immunomodulators and biologics.
How does ulcerative colitis develop?
Ulcerative colitis typically begins in the rectum and can either remain confined to it, which is called ulcerative proctitis, or extend proximally, sometimes to the entire colon. In very rare cases, the entire colon is affected.
The inflammation in ulcerative colitis affects the mucosa and submucosa of the intestine, with a clear boundary between normal and affected tissue. Only in severe cases is the muscularis also affected. At the beginning of the disease, the mucosa is reddened, fine-grained and fragile, with a loss of the normal vascular structure and often isolated bleeding areas. In severe cases, large ulcers are present in the mucous membrane, accompanied by copious purulent discharge. In the midst of the damaged mucosa, islands of relatively normal or hyperplastic inflammatory mucosa (pseudopolyps) protrude. Fistulas and abscesses are not typical features of this disease.
What are the symptoms and signs of ulcerative colitis?
Patients typically experience episodes of bloody diarrhea of varying intensity and duration, alternating with symptom-free periods.
An attack often begins gradually with an increased urge to defecate, mild lower abdominal cramps and blood and mucus in the stool. Sometimes such episodes occur after infections such as amoebiasis or bacterial dysentery. If the ulcerations are confined to the rectosigmoid, the stool may be normal or hard, but rectal discharges of mucus mixed with red and white blood cells occur simultaneously or between bowel movements. Systemic symptoms are usually mild or absent. As the ulcerations spread proximally, the stool becomes softer and the patient often has more than 10 bowel movements per day, often accompanied by severe cramps and painful rectal urges, especially at night. The stool may be watery, contain mucus and often consist entirely of blood and pus, and systemic symptoms and signs are more common in extensive ulcerative colitis, including malaise, fever, anemia, loss of appetite and weight loss. Extraintestinal manifestations of inflammatory bowel disease (IBD), especially joint and skin complications, are more common when systemic symptoms are present.
What is toxic colitis?
Toxic colitis occurs when the ulcerations expand transmurally and lead to localized intestinal obstruction or peritonitis. Within a few hours or days, the colon loses its muscle tone and begins to dilate. Toxic or fulminant colitis initially manifests as sudden onset of severe diarrhea, fever up to 40°C, abdominal pain, signs of peritonitis and marked toxic symptoms.
It is important to note that the terms "toxic megacolon" and "toxic dilatation" should be avoided, as the toxic inflammatory condition and its complications can occur without obvious megacolon (defined as a transverse colon greater than 6 cm in diameter during an exacerbation).
Toxic colitis is a medical emergency that usually occurs spontaneously during the course of very severe colitis, but can occasionally be triggered by the use of opioids or anticholinergic, antidiarrheal medications. Intestinal perforation can occur, which significantly increases the mortality rate.
How is ulcerative colitis diagnosed?
The suspected diagnosis of ulcerative colitis is based on the typical symptoms and findings, especially in the presence of extraintestinal manifestations or a history of previous similar attacks. Ulcerative colitis must be differentiated from Crohn's disease and, more importantly, from other causes of acute colitis (e.g. infection, ischemia in older patients).
Stool cultures for intestinal pathogens must be taken in all patients and Entamoeba histolytica should be excluded by examining fresh stool samples. If amoebiasis is suspected epidemiologically or on the basis of a travel history, appropriate serological titer tests and biopsies should be obtained. A history of previous antibiotic therapy or recent hospitalization should be followed by stool testing for Clostridioides difficile (formerly Clostridium difficile) toxin. Patients at risk should be tested for HIV, gonorrhea, herpes virus, chlamydia and amoebiasis. In immunocompromised persons, opportunistic infections (e.g. cytomegalovirus, Mycobacterium avium intracellulare) or Kaposi's sarcoma must be considered. In women taking oral contraceptives, the presence of contraceptive-induced colitis is a possibility; it usually resolves spontaneously after discontinuation of the hormones. Stool tests for lactoferrin and fecal calprotectin can be helpful in differentiating IBD from functional diarrhea.
A sigmoidoscopy or colonoscopy should be performed. It allows visual confirmation of colitis, direct sample collection of stool or mucus for culture, microscopic examination and biopsy of the affected region. Although visual findings and biopsies may not lead to a diagnosis, as there is a lot of overlap between the different types of colitis, acute, self-limiting, infectious colitis can usually be distinguished histologically from chronic idiopathic ulcerative colitis or Crohn's disease. Severe perianal disease, recess of the rectum, absence of bleeding and asymmetric or segmental involvement of the colon are more suggestive of Crohn's disease than ulcerative colitis. Colonoscopy is not usually required initially, but should be performed electively if the inflammation has spread proximally beyond the sigmoidoscope's field of view.
Laboratory tests should be directed towards the detection of anemia, hypoalbuminemia and electrolyte disturbances. Liver tests should be obtained: Elevated alkaline phosphatase and gamma-glutamyltranspeptidase are suspicious for the presence of primary sclerosing cholangitis. Perinuclear antineutrophil cytoplasmic antibodies are relatively specific (60-70%) for ulcerative colitis. Anti-Saccharomyces cerevisiae antibodies are relatively specific for Crohn's disease. However, these tests cannot reliably differentiate between the two diseases and are not recommended for routine diagnostics. Other possible pathological laboratory findings are leukocytosis, thrombocytosis and elevated acute phase proteins (e.g. erythrocyte sedimentation rate, C-reactive protein).
X-ray examinations are not diagnostic, but occasionally show pathological changes. Survey radiographs of the abdomen may show mucosal edema, loss of haustration, and absence of formed stool in the affected bowel; however, the yield is low and they are not routinely recommended. Barium enemas show similar changes, although more marked, and may additionally show ulceration, but should not be used during the acute phase. After several years of disease progression, a shortened rigid colon with an atrophic or pseudopolypoid mucosa is often seen. Radiographic findings with thumbprint signs and segmental distribution are more suspicious of intestinal ischemia or possibly Crohn's colitis than ulcerative colitis.
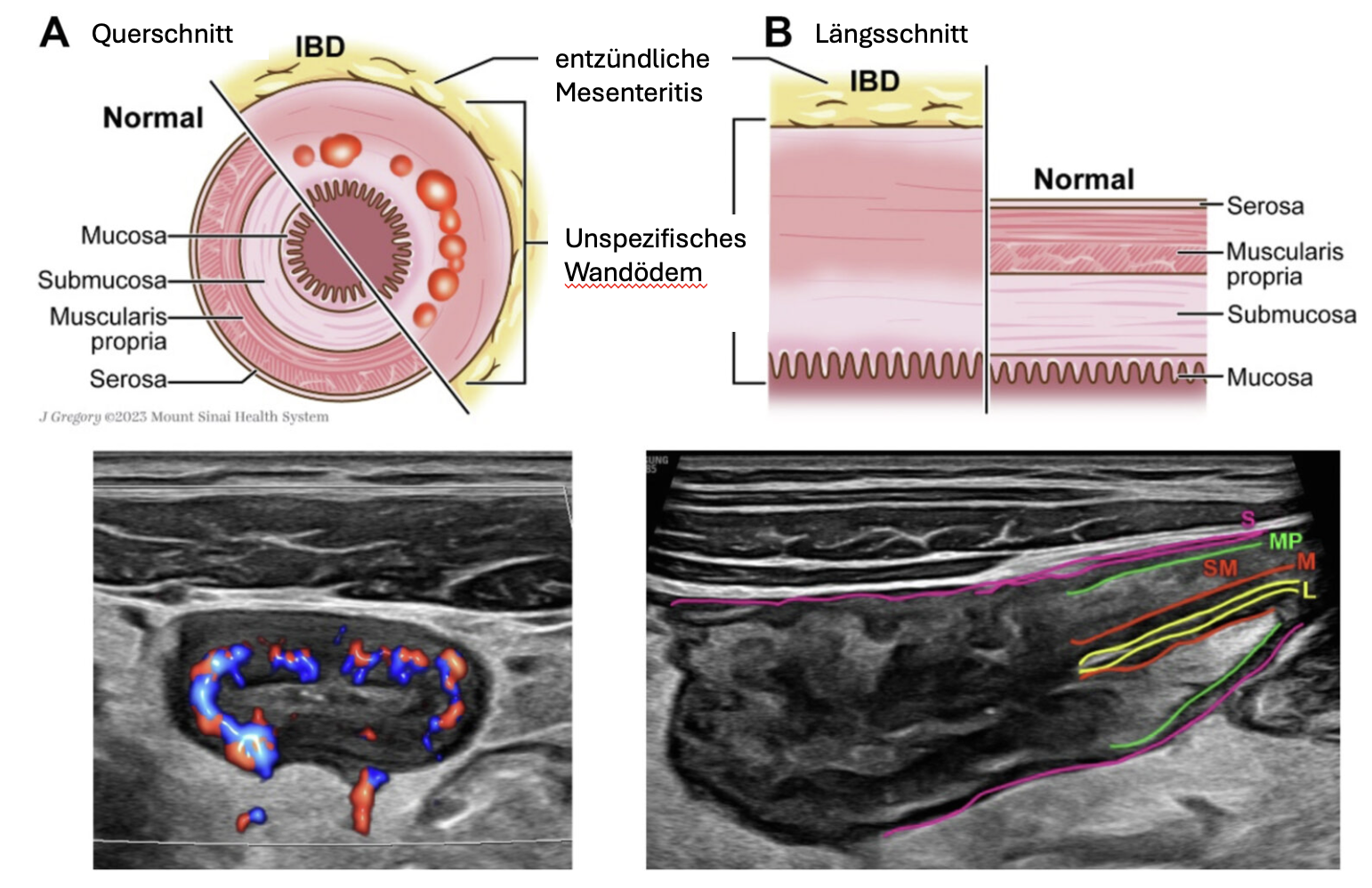
High-resolution ultrasound in ulcerative colitis.
Recurrent symptoms
Patients with known underlying disease and recurrent typical symptoms do not always require extensive examinations. Depending on the duration and severity of the symptoms, a sigmoidoscopy or colonoscopy can be performed and the complete blood count determined. If the possible recurrence is not actually typical or if there is an exacerbation after a long period of remission or if contact with a pathogen is suspected, after antibiotics or simply whenever the clinician suspects something else, cultures should be taken and examined for eggs and parasites and for C. difficile toxin.
Acute pain attacks
Patients require thorough hospitalization during severe episodes of inflammation. A supine and standing abdominal survey should be performed. These may show a megacolon or intraluminal gas accumulation in an extensive, continuous and paralyzed segment of the colon - as a result of loss of muscle tone. Barium enema should be avoided because of the risk of perforation, but careful sigmoidoscopy is usually advisable to assess severity and rule out infection. Blood status, platelet count, erythrocyte sedimentation rate, C-reactive protein, electrolytes and albumin should be determined; aspartate aminotransferase, alanine aminotransferase, and blood group and cross match are also indicated in cases of severe bleeding.
The patient must be closely monitored because of the risk of progressive peritonitis and perforation. Percussion should be performed over the liver because loss of hepatic attenuation may be the first sign of free perforation, especially in a patient whose peritoneal signs may be suppressed by high-dose corticosteroid therapy. Abdominal X-rays are performed every two days to assess the progression of dilatation of the colonic loops and to detect possible free or intramural air, CT is more sensitive in detecting extraluminal air or pericolic abscess.
What are strategies for managing ulcerative colitis?
Drug therapy: Your doctor may prescribe medications to control inflammation and relieve symptoms. Dietary optimization: An adjusted diet, possibly in conjunction with a dietitian, can help identify trigger foods and minimize discomfort. stress management: Techniques such as meditation, yoga and stress reduction can help to reduce symptoms. Regular medical check-ups: Continued monitoring by a medical specialist is crucial to assess disease progression and adjust the treatment plan.
When to seek medical help? Seek medical attention immediately if you notice symptoms such as persistent diarrhea, blood in the stool or severe abdominal pain. Early diagnosis and treatment can have a positive effect on the course of the disease.
- Dietary treatment and loperamide (except for acute severe attacks) to relieve symptoms
- 5-aminosalicylic acid (5-ASA)
- Corticosteroids and other medications, depending on the symptoms and severity
- Antimetabolites, biologics, a Janus kinase inhibitor or sphingosine-1-phosphate (S1P) receptor modulator
- Occasionally surgery
General treatment measures
Avoiding raw fruit and vegetables limits damage to the inflamed colonic mucosa and can improve symptoms. Dairy-free diet may be helpful, but need not be continued if no beneficial effect is observed. Loperamide 2 mg orally 2 to 4 times daily is indicated for relatively mild diarrhea, higher doses (4 mg in the morning and 2 mg after each bowel movement) should be used for more severe diarrhea. Overall, antidiarrheal drugs must be used with extreme caution in severe cases because they can cause toxic dilatation. All patients with inflammatory bowel disease should be advised to take adequate amounts of calcium and vitamin D.
Routine health care measures (e.g. vaccinations, cancer screening) should be emphasized.
Mild left-sided disease
Patients with mild to moderate ulcerative proctitis or proctosigmoiditis that does not extend proximally beyond the sigmoid colon are treated with 5-ASA (mesalamine) by enema once or twice daily, depending on severity. Suppositories are effective for more distal infestations and are generally preferred by patients. Corticosteroid and budesonide enemas are somewhat less effective, but are used if 5-ASA is ineffective or is not tolerated. When remission is achieved, the dose is slowly tapered to maintenance levels. Oral 5-ASA drugs theoretically have the additional advantage of reducing the likelihood of proximal extension of the disease.
Moderate or extensive disease
Patients with inflammation proximal to the sigmoid colon or left-sided disease that does not respond to topical agents should receive a biologic agent (infliximab, adalimumab, golimumab, ustekinumab, vedolizumab) with or without immunomodulators (azathioprine or 6-mercaptopurine). Sometimes high-dose corticosteroids are added to induce remission in patients with moderate to severe ulcerative colitis. A combination of immunomodulators and anti-tumor necrosis factor therapy is sometimes helpful. Finally, a trial of the Janus kinase inhibitor (tofacitinib or upadacitinib) or a trial of the sphingosine-1-phosphate receptor modulator ozanimod may be considered in some patients.
Severe disease
Patients with > 10 bloody bowel movements per day, tachycardia, high fever, or severe abdominal pain must be hospitalized to receive a biologic agent and/or high-dose IV corticosteroids. Intravenous fluid replacement and blood transfusions will be administered as needed to correct dehydration and anemia. The patient must be closely monitored due to the risk of developing toxic colitis. Patients who can tolerate food should eat. Sometimes parenteral hyperalimentation is used for nutrition, but this is worthless as a primary therapy.
Patients who do not respond to biologics and/or corticosteroids within 3-7 days should be considered for intravenous cyclosporine or surgery. Patients who do not respond to a corticosteroid regimen are switched to prednisone 40 mg orally once daily within one week, which can be gradually reduced at home depending on clinical response. Patients who have started ciclosporin IV and respond to therapy are switched to ciclosporin orally and azathioprine or 6-mercaptopurine at the same time. Oral application of cyclosporine is continued for about 3-4 months, during which time corticosteroids are discontinued and cyclosporine levels are closely monitored. Some clinicians recommend prophylaxis against Pneumocystis jirovecii pneumonia during the interval of overlapping treatment with corticosteroids, cyclosporine and antimetabolites. Tacrolimus, an immunosuppressive agent also used in transplant recipients, appears to be as effective as ciclosporin and may be considered for patients with severe or refractory ulcerative colitis who do not require hospitalization. The trough levels should be maintained between 10 and 15 ng/ml (12 to 25 nmol/l).
Maintenance therapy
After successful treatment of an inflammatory episode, corticosteroids are tapered according to the clinical response and eventually discontinued, as they are not effective as maintenance therapy. Patients should receive 5-ASA medications indefinitely, orally or rectally, depending on the location of the disease, because interruption of maintenance therapy often leads to relapse. The dosing intervals for rectal preparations can be slowly extended to every 2nd or 3rd day. There is sufficient evidence that a combination of oral and rectal therapy is significantly more effective than either therapy alone.
Patients who cannot be weaned off corticosteroids should receive thiopurines (azathioprine or 6-mercaptopurine), methotrexate, biologics, Janus kinase inhibitors, the sphingosine-1-phosphate receptor modulator ozanimod, or a combination of these drugs.
Patients who have started induction therapy with infliximab should also use this medication for maintenance therapy
Surgery
Approximately one third of patients with severe ulcerative colitis ultimately require surgery. Total proctocolectomy is curative: life expectancy returns to normal and the risk of colon cancer is significantly reduced. However, some studies report that up to 25% of patients later develop inflammation of the small intestinal mucosa, which is indicative of Crohn's disease; this can occur many years after the operation. After total proctocolectomy with ileum pouch-anal anastomosis (IPAA), a small risk of dysplasia or cancer remains in the rectal-anal junction area and also in the ileum pouch. After proctocolectomy with ileostomy or IPAA, the quality of life is improved; however, new quality-of-life challenges are created.
Emergency colectomy is indicated in cases of massive bleeding, fulminant toxic colitis or perforation. Subtotal colectomy with ileostomy and closure of the rectosigmoid (Hartmann procedure) or mucosal fistula is usually the procedure of choice because the majority of critically ill patients cannot tolerate a more extensive surgical procedure. The rectosigmoid stump can be removed electively at a later date or used for an ileoanal anastomosis with a pouch. An intact rectal stump should not be left indefinitely due to the risk of disease activation or malignant transformation.
Elective surgery is indicated for proven carcinoma, symptomatic strictures, growth retardation in children or most commonly for refractory chronic disease with incapacitation or corticosteroid dependence. Severe colitis-dependent extraintestinal manifestations (e.g. pyoderma gangraenosum) can nowadays be better controlled by intensive medical procedures and are only rarely indications for surgical intervention.
The elective procedure of choice for patients with normal sphincter function is reconstructive proctocolectomy with ileoanal anastomosis. This procedure creates a reservoir in the pelvis or a pouch of distal ileum that is connected to the anus. The intact sphincter typically maintains continence with 4-9 stools/day (including 1 or 2 at night).
Pouchitis is an inflammatory reaction that occurs after restorative proctocolectomy with IPAA+ in approximately 50% of patients. The risk of pouchitis appears to be higher in patients with primary sclerosing cholangitis than in patients with preoperative extraintestinal manifestations and possibly in patients with high preoperative serologic titers of perinuclear antineutrophil antibodies and other inflammatory bowel disease biomarkers. The development of pouchitis is probably related to bacterial overgrowth and is treated with antibiotics (e.g. quinolone). Probiotics may be protective. The majority of cases of pouchitis can be kept under control, but 5-10% prove refractory to all drug measures and require conventional (Brooke) ileostomy. For a minority of patients who are elderly, well established and have a higher lifestyle, have low sphincter tone or cannot tolerate frequent bowel movements, or are simply unable or unwilling to manage the consequences of frequent or chronic pouchitis, Brooke ileostomy remains the method of choice.
Ileitis following IPAA or end ileostomy is treated in the same way as other forms of IBD. Surgical interventions are rarely required.
In any case, the physical and emotional stress of any form of colon resection should be taken into account and care should be taken to ensure that the patient receives all instructions and the necessary medical and psychological support before and after the operation.
Conclusion: Ulcerative colitis can be challenging, but with a comprehensive approach and professional care, quality of life can be improved. Learn more about proven strategies for managing ulcerative colitis and take an active role in your health.
Goals of ulcerative colitis treatment
- Alleviate symptoms
- Soothe or heal the mucous membrane
- Prevent relapses
- Achieve steroid-free remission
- Avoid surgery
- Improve quality of life
monitoring plan for patients
The guideline recommends
- Stool calprotectin every 3-6 months or when symptoms occur
- CRP, blood values
regularly - Sigmoidoscopy/colonoscopy
for therapy adjustment, relapse, or every 1-3 years - Ultrasound
- Screening for anxiety/depression
annually
Current therapy recommendations for biologics and small molecules (moderate to severe ulcerative colitis).
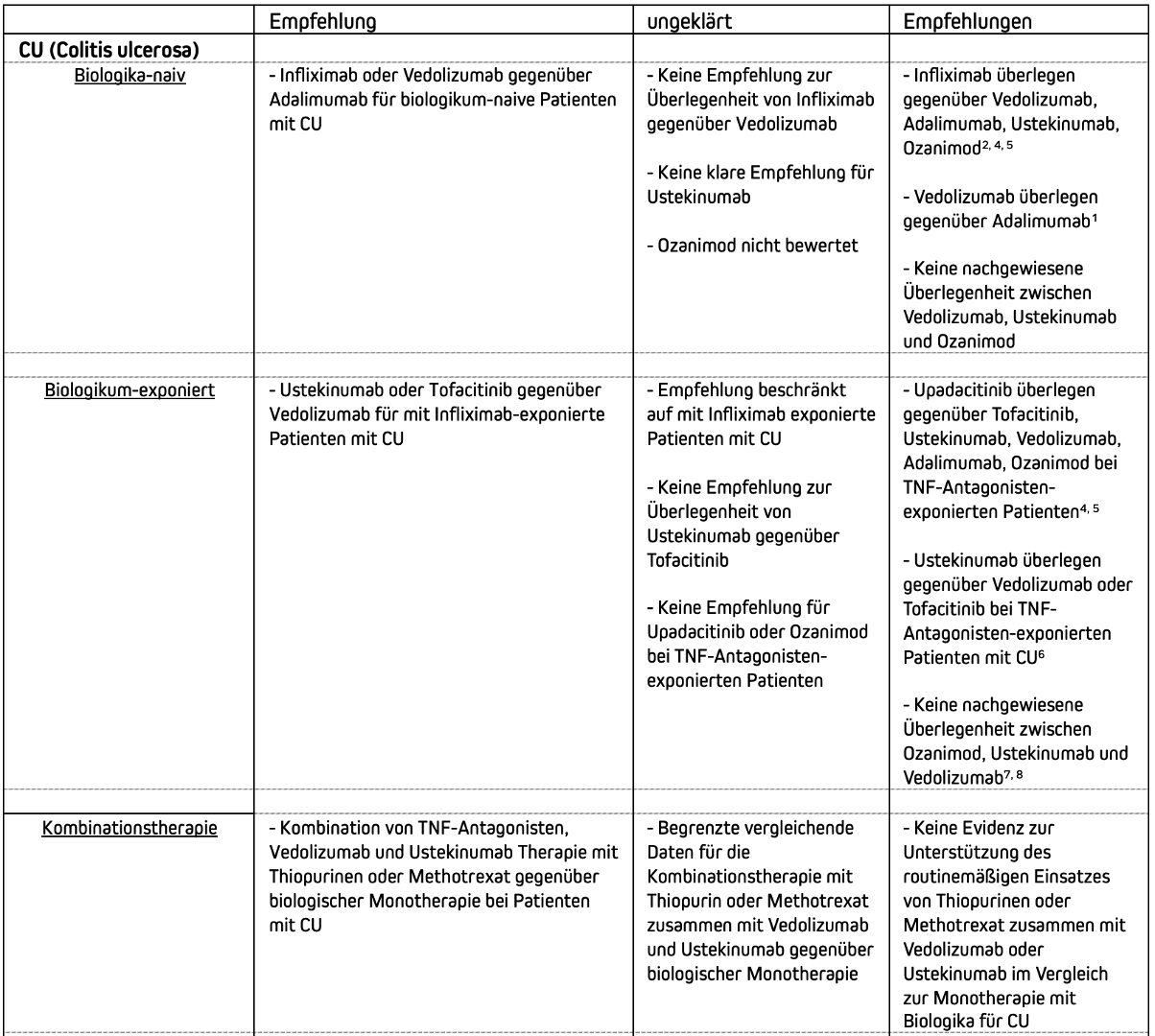
Treatment recommendations for biologics and small molecules (moderate to severe ulcerative colitis)
SOURCES:
1. Sands BE, Peyrin-Biroulet L, Loftus EV Jr, et al. Vedoli-zumab versus adalimumab for moderate-to-severe ulcerative colitis. N Engl J Med 2019;381:1215-1226.
2. Narula N, Wong ECL, Marshall JK, et al. Comparative efficacy for infliximab vs vedolizumab in biologic naive ulcerative colitis. Clin Gastroenterol Hepatol 2022; 20:1588-1597 e3.
3. Narula N, Wong ECL, Dulai PS, et al. Comparative effectiveness of biologics for endoscopic healing of the ileum and colon in Crohn's disease. Am J Gastroenterol 2022;117:1106-1117.
4. Lasa JS, Olivera PA, Danese S, et al. Efficacy and safety of biologics and small molecule drugs for patients with moderate-to-severe ulcerative colitis: a systematic review and network meta-analysis. Lancet Gastroenterol Hepatol 2022;7:161-170.
5. Burr NE, Gracie DJ, Black CJ, et al. Efficacy of biological therapies and small molecules in moderate to severe ulcerative colitis: systematic review and network meta-analysis. Gut 2022;71:1976-1987.
6. Kochhar GS, Desai A, Farraye FA, et al. Efficacy of bio-logic and small molecule agents as second-line therapy after exposure to TN inhibitors in patients with ulcerative colitis: a propensity-matched cohort study. Aliment Pharmacol Ther 2023;58:297-308.
7. Yzet C, Diouf M, Singh S, et al. No benefit of concomitant immunomodulator therapy on efficacy of biologics that are not tumor necrosis factor antagonists in patients with inflammatory bowel diseases: a meta-analysis. Clin Gastroenterol Hepatol 2021;19:668-679.
8.Hu A, Kotze PG, Burgevin A, et al. Combination therapy does not improve rate of clinical or endoscopic remission in patients with inflammatory bowel diseases treated with vedolizumab or ustekinumab. Clin Gastroenterol Hepatol 2021;19:1366-1376.
9. Barberio B, Gracie DJ, Black CJ, et al. Efficacy of biological therapies and small molecules in induction and maintenance of remission in luminal Crohn's disease: systematic review and network meta-analysis. Gut 2023; 72:264-274.
10. Kirchgesner J, Desai RJ, Schneeweiss MC, et al. Decreased risk of treatment failure with vedolizumab and thiopurines combined compared with vedolizumab mon-otherapy in Crohn's disease. Gut 2022;71:1781-1789.
11. Wong ECL, Dulai PS, Marshall JK, et al. Comparative efficacy of infliximao vs ustekinumab for maintenance of clinical response in biologic naïve Crohn's disease. Inflamm Bowel Dis 2023;29:1015-1023.
12. American College of Gastroenterology, Management of Crohn's Disease in Adults, 06, 2025.